Hot Dip Galvanizing
Ask Professor Zinc
November 2017
Q: "Why does hot dipping make welds seem to pop up?"

Answer:
A. The answer is most likely the difference between the chemistry of the steel and the weld metal. Galvanized coating thickness primarily depends on the silicon content of the iron or steel part. The major difference between the weld metal and the structural steel is the amount of silicon in the weld rod. Excessive silicon in the weld filler material can accelerate the growth of the hot-dip galvanized coating. Because some weld rod metal contains nearly 1% silicon, the difference between the coating thickness on the weld metal and the surrounding structural steel can be significant. Excessive silicon in the weld material to be galvanized causes an accelerated formation of the zinc-iron layers that make up the hot-dip galvanized coating, greatly increasing coating weight.
When the fabricated structure is immersed in the zinc bath long enough to achieve a coating that meets the minimum thickness of the galvanizing standards (such as ASTM A 123/ A 123M, Standard Specification for Zinc (Hot-Dip Galvanized) Coatings on Iron and Steel Products), the coating on the high-silicon weld metal can be more than two-times thicker than the surrounding coating. This thick coating on the weld detracts from the appearance of the fabricated structure and increases the possibility of the zinc coating becoming damaged in the weld area with further handling of the assembly or part.
For typical welding processes, such as shielded metal arc welding (SMAW), submerged arc welding (SAW) and flux-cored arc welding (FCAW), there are weld rod materials that will not cause excessively thick coatings.
Thanks for the question! We hope you find the answer helpful.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
September 2017
Q: "Can you dip something measuring 9' 2" wide x 15' long x 3' thick? It is made of I-beams and channels. Here is a photo."

Answer:
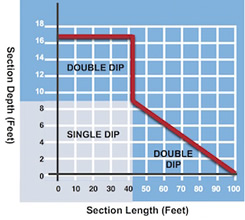
Absolutely. When a fabrication is too large for a single immersion, it is often possible to galvanize it by progressive dipping, immersing one end of the work at a time in the galvanizing bath. Galvan Industries’ hot dip galvanizing kettle is 42′ long x 4’6″ wide x 8’6″ deep. With “progressive” or “double dipping”, our large bath size will allow us to hot dip galvanize almost any structure for greatly reduced maintenance costs and extended life.
The galvanizing capacity chart below indicates the maximum length of structural sections capable of being galvanized at our facility. The depth of section is indicated along the vertical axis and the corresponding maximum length is shown on the horizontal axis.
Note: These sizes are theoretical maximums for length and depth. Please contact Galvan Industries on fabrications that approach the extreme limits shown.
Because hot-dip galvanizing is a total immersion process, it is always a good idea to verify kettle constraints with your galvanizer in advance when dealing with a large items like this one.
Thanks for the great question! We hope you found the answer helpful.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
July 2017
Q: "What is the best arrangement of vent holes in a sealed rectangular tube that is 10 inches by six inches? Length varies."
Answer:
ASTM A385/A385M specifies that a minimum vent opening of 30% of the cross-sectional area of the tubular structure must be included on each end of the capped tube. This is so that no air or solutions are trapped inside the assembly during galvanizing. In this case, the cross-sectional area is 60 sq. in., which means the vent openings must equal 18 sq. in. (30%).
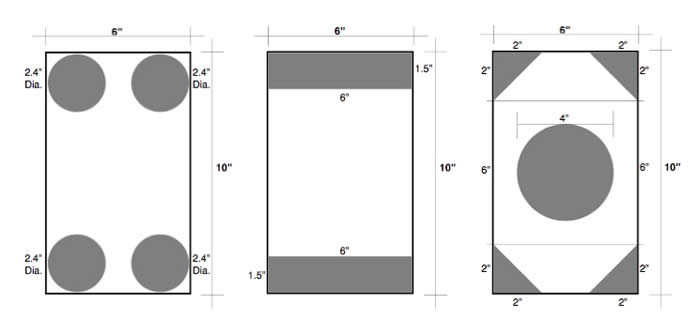
The diagrams here show three different ways to achieve the 30% vent opening. All three meet the requirements of the spec. The best arrangement is the one that works best for your situation or application.
Thanks for the great question! We hope you found the answer helpful.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
May 2017
Q: "What is hot dip galvanizing CLASS B-2?"
Answer:
Class B-2 is a materials classification from ASTM A153 for hardware products such as castings, fasteners and miscellaneous threaded objects that are centrifuged, spun, or otherwise handled to remove excess zinc. Class B is refers specifically to rolled, pressed, and forged articles. B-2 identifies such articles under 3/16 inch (4.76mm) in thickness and over 15 inches (381mm) in length.
The requirements for ASTM A153/A153M are very similar to those for ASTM A123/A123M, except for the addition of threaded products and embrittlement requirements.
ASTM A153/A153M Requirements
• Finish – continuous, smooth, uniform
• Embrittlement – high tensile strength fasteners (>150ksi) and castings can be subject to embrittlement
• Appearance – free from uncoated areas, blisters, flux deposits and gross dross inclusions as well as having no heavy zinc deposits that interfere with intended use
• Adherence – the entire coating should have a strong adherence throughout the service life of hot-dip galvanized steel
• Threaded Products – areas with threads are not subject to the coating thickness requirement
• Coating Thickness/Weight – depends on the material category and steel thickness, as listed in Table 3 of the specification. For Class B-2, the requirements are as follows:
| Weight (Mass) of Zinc Coating, | Coating Thickness, mils | |||
| oz/ft2 (g/m2) of Surface, minimum | (microns), Minimum | |||
| Class of Material | Average of Specimens Tested | Any Individual Specimen | Average of Specimens Tested | Any Individual Specimen |
| B-2 | 1.5 (458) | 1.25 (381) | 2.6 (66) | 2.1 (53) |
For more information, visit the Galvanizing Standards section of the AGA web site, www.galvanizeit.org.
Thanks for the great question! We hope you found the answer helpful.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
March 2017
Q: "We have a supplier of an endfitting overseas that is having trouble galvanizing the inside of a cast fitting due to a deep pocket in the fitting. I have attached a few pictures. Could you explain the difficulty of getting the galvanization to work in this area?"
Answer:


A.There are two possible explanations for the bare spots inside the castings. The first is that they were not properly cleaned before dipping. Castings must be abrasive cleaned by shot blasting prior to the galvanizing process. If there is any sand or other residue left from the casting process inside the part, it will not galvanize correctly. At Galvan, we blast clean every casting prior to galvanizing.
The second possible explanation is the formation of an air pocket inside the part. If this part is not hung at exactly the right angle when it is being dipped, an air pocket will form at the back of casting and it will not get full coverage of zinc. The air will not allow the zinc to reach the back of the part.
A fix could be an added hole through the back of the part to allow the air to escape. However, if the purpose of this part is to stop the flow of a liquid or gas, adding a hole would create another problem.
If that is the case, the answer to the problem is greater care in cleaning to eliminate sand inclusions and greater care in dipping to ensure that air inside the casting is allowed to escape.
Regardless of what caused the problem, these parts as shown should be cleaned and repaired, or rejected, stripped, and regalvanized.
Thanks for your question and good luck in resolving this issue.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
January 2017
Q: "Does the cost of galvanizing really pay off for transportation infrastructure projects like bridges and airports?"
Answer:
A. Airports, bridges, highways and mass transit systems are major public investments. It is critical to protect these large investments with a sustainable, durable, maintenance-free corrosion protection system that will withstand the effects of constant rough usage and environmental exposure. Hot dip galvanizing the steel used in infrastructure projects from rebar and structural steel to guard rail and sign posts will lead to significant savings in maintenance and repair costs. The extended the life of projects in question will also save or delay replacement costs.
The annual direct cost of corrosion for highway bridges alone is estimated to be $6.3 to $10.15 billion, a cost that has to be paid by state and federal taxes. Building every new bridge – structural steel or reinforced concrete – with galvanized steel would keep those costs from rising further and reverse them in the future as older bridges are replaced. Optimizing the return on an infrastructure investment means building things to last as long as possible, safely and maintenance-free. Given that, galvanizing definitely pays off.
Happy New Year and thanks for the question. We hope you find the answer helpful.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
November 2016
Q: "What are the basic requirements for drain and vent holes in hollow fabrications?”
Answer:
A. In the hot dip galvanizing process, steel fabrications are lowered into a bath of molten zinc at a temperature around 860 degrees F. The high temperature will rapidly raise the internal pressure inside hollow structures (up to 3600 psi) causing them to rupture or explode.
Therefore, all hollow fabrications being galvanized must allow for the venting and draining of air, moisture, and molten zinc for safety reasons.
Venting and drainage also affect lifetime performance of the galvanized structure, ensuring that all interior and exterior surfaces are properly cleaned and galvanized to protect them from corrosion, inside and out.
The size and location of the holes vary depending on the product, but they critical to achieving successful and safe galvanizing. Because items being galvanized are immersed in and withdrawn at an angle from cleaning solutions, flux solutions and molten zinc, vent holes should be located at the highest point and drain holes at the lowest point as mounted during the galvanizing process.
The size of holes also has an impact. Larger holes allow faster flow of zinc in and out of the article, making immersion and withdrawal easier. This will result in a better quality finish.
A variety of fabricated steel products require vent and drain holes. Galvan has produced a Design Guide for Hot Dip Galvanizing with examples of good venting and draining from the American Galvanizing Association. It is available as a download from our Web site at http://www.galvan-ize.com/galvanizing.asp. You can also request a printed copy by email from sales@galvan-ize.com.
Thanks for the question. We hope you find the answer helpful.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
August 2016
Q: "We received some steel galvanized pipe (thick wall 1/2 inch) that we are supposed to brush blast and coat. It doesn't handle the brush blast without heavy peeling. I checked the thickness of the zinc. It ranges from 6 mils to well over 100 mils. The thin areas handle the blast well but the thick just swells from the friction and pops off. Is this acceptable?"
Answer:
The primary cause for peeling or delamination on heavy steel parts is the time it takes for them to cool off after dipping in the zinc bath.
During the hot-dip galvanizing process, steel is heated to approximately 830 F (443 C) by immersing it in a molten zinc bath. The bath is maintained at that temperature.
While immersed in the kettle, the iron in the steel reacts with the zinc to form a series of zinc-iron intermetallic alloy layers. Once the item being dipped reaches the same temperature as the bath, it is withdrawn. Excess zinc is removed by draining or other methods, but the metallurgical reaction will continue after withdrawal from the bath, as long as the article remains near bath temperature.
Large, thick galvanized parts may take a long time to cool and continue to form zinc-iron layers after they have been removed from the galvanizing kettle.
This continued coating formation can leave create a void between the top two layers of the galvanized coating. If there are many voids formed, the top layer of zinc can separate from the rest of the coating and peel off, leaving a rough textured gray finish.
If the remaining coating still meets the minimum specification requirements for thickness, then the part is still acceptable. If the coating does not meet the minimum specification requirements, then the part should be rejected and regalvanized.
However, if delamination occurs as a result of fabrication or processing after galvanizing, such as blasting before painting, then the galvanizer is not responsible for the problem.
Thanks for the question! I hope the answer is helpful.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
June 2016
Q: "Is electroplating per ASTM B633 equal to hot dip galvanizing per ASTM A385?"
Answer:
No, they are not equal. Hot dip galvanizing creates a thicker and harder zinc coating with a service life as much as 20 times longer than a B633 coating.
Actually B633 is the Standard Specification for Electrodeposited Coatings of Zinc on Iron and Steel. In general terms, the zinc coating is thin, up to a maximum thickness of 1 mil (25 μm), and mechanically bonded to the surface with a hardness of about a third to a half that of most steels. The specification, ASTM B633, lists four classes of zinc-plating: Fe/Zn 5, Fe/Zn 8, Fe/Zn 12 and Fe/Zn 25 where the number indicates the coating thickness in microns (μm). Most coatings are less than a half of a mil in thickness and are intended for indoor and/or non critical applications.
ASTM A385 is not actually a coating specification but the Standard Practice for Providing High-Quality Zinc Coatings (Hot-Dip), and is commonly referred to as the “design” spec for galvanizing.
ASTM A123 Standard Specification for Zinc (Hot-Dip Galvanized) Coatings on Iron and Steel
Products and ASTM A153 Standard Specification for Zinc Coating (Hot-Dip) on Iron and Steel Hardware, are more likely the specifications you want to compare to.
Hot dip galvanized structural material will range from 45 μm to 100 μm as a minimum (1.8 to 3.9 mil) and hot dipped fasteners will range from 45 μm to 86 μm (1.7 to 3.4 mil), depending upon the type of material being coated.
Since zinc coating performance is linear and is based on coating thickness and conditions of exposure, a two mil coating will last twice as long as a one mil coating in the same environment. You can see from the relative coating thicknesses of electrogalvanizing and hot dip galvanizing that they are NOT equal. Hot dip galvanizing will have a service life that may be as much as 20 times the life of a B633 coating in the same application.
Thanks for the great question.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
April 2016
Q: "We are supplying cable trays to a project with a sheet steel thickness of 2mm and 2.5mm. Can the same be coated with 85 micron coating thickness? The standards specify coating between 45 micron and 55 microns."
Answer:
Achieving a consistent 85-micron coating thickness might be difficult in your application. Galvanizing is an alloy that forms when the prepared steel surface is immersed in a bath of molten zinc. The galvanizing bath temperature is 840-845 degrees F, and the steel remains in the bath until the thickest part of the steel fabrication reaches the temperature of the bath. When the steel core reaches the bath temperature the reaction stops and the steel has been galvanized.
The coating reaction begins while the steel is heating and stops when it reaches bath temperature. Thin or light structures reach bath temperature faster than thick or heavy sections, limiting the thickness the coating can achieve.
There are other factors that can affect the coating, such as unusual steel chemistry, but for this discussion, we’ll focus on standard mild carbon structural material.
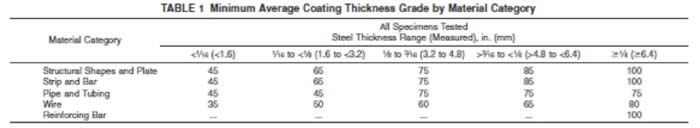
Please refer to ASTM A123 for complete details on the minimum thickness values for different materials. The categories listed in ASTM A123 are Structural Shapes and Plates, Strip and Bar, Pipe and Tubing, Wire, and Reinforcing Bar. The information below relates to Structural Shapes and Plates. The minimum standard coating thickness is shown below the measured steel thickness.
Steel Thickness Range (Measured), in. (mm)
<1⁄16 (<1.6) 1⁄16 to <1⁄8 (1.6 to <3.2) 1⁄8 to 3⁄16 (3.2 to 4.8) >3⁄16 to <1⁄4 (>4.8 to <6.4) ≥1/4 (≥6.4)
45 microns 65 microns 75 microns 85 microns 100 microns
1.8 mils 2.6 mils 3.0 mils 3.3 mils 3.9 mils
Based on this information, your specified standard of 45 to 55 microns of galvanizing thickness sounds reasonable for 2 to 2.5-mm material, but keep in mind that ASTM 123 is a specification for MINIMUM coating thickness. Galvanizing will normally meet or exceed these minimums in every case. However, to get to 85 microns, even starting at the upper end of your range, you’d need to exceed the minimum by more than 50%. At the lower end of the range, you’d need to nearly double the specified thickness. It may be possible, but thin materials work against thick coatings in the galvanizing process.
Good luck! I hope this answer helps!
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
February 2016
Q: "I have an issue with hot dip galvanizing for small parts. After centrifuging, galvanizing material separates from the metal. Steel is AISI 1140. How can we overcome this issue?"
Answer:
Steel chemistry is a likely cause of the problem. For AISI 1140, the approximate percentage of silicon is 0.4%. This is significantly higher than .22% which is the maximum silicon percentage recommended for normal or typical galvanized coating growth. AISI 1140 also has a higher percentage of carbon than is recommended. If the coating growth due to such trace elements is excessive, the coating can be susceptible to flaking.
Trace elements affect the galvanizing process as well as the structure and appearance of the galvanized coating. Steels with trace elements outside of the recommended ranges are known in the galvanizing industry as highly reactive steel, and may produce an atypical coating composed entirely, or almost entirely, of zinc-iron alloy layers.
In appearance, the atypical galvanized coating may have a matte gray and/or rougher finish. The free zinc layer present on typical coatings imparts a shinier finish to a galvanized coating. The zinc-iron alloy coating also tends to be thicker than a typical galvanized coating. In the rare situation where the coating is excessively thick, there is the possibility of diminished adhesion (flaking) under external stress from temperature or impact.
The solution may be to use a different type of steel that contains less silicon and other elements that may cause the problems in galvanizing. Here are some guidelines.
Carbon less than 0.25%
Phosphorus less than 0.04%
Manganese less than 1.35%
Silicon levels less than 0.04% or between 0.15% - 0.22%
Thank you for your question. We hope you found the answer helpful.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
December 2015
Q: "Can we ream or re-drill holes on galvanized steel members?"
Answer:
You can certainly ream or drill holes after galvanizing. However the drilled areas must be within the guidelines below to certify an ASTM A123 coating. You will want to touch up as required per ASTM A780 specification.
From ASTM A123:
"Surfaces that remain uncoated after galvanizing shall be renovated in accordance with the methods in Practice A780 unless directed by the purchaser to leave the uncoated areas untreated for subsequent renovation by the purchaser.
6.2.1 Each area subject to renovation shall be 1 in. [25 mm] or less in its narrowest dimension.
6.2.2 The total area subject to renovation on each article shall be no more than 1⁄2 of 1 % of the accessible surface area to be coated on that article, or 36 sq. in. per short ton [256 cm2 per metric ton] of piece weight, whichever is less.
NOTE 5—Inaccessible surface areas are those which cannot be reached for appropriate surface preparation and application of repair materials as described in Practice A780. Such inaccessible areas, for example, would be the internal surfaces of certain tanks, poles, pipes, tubes, and so forth.
6.2.3 The thickness of renovation shall be that required by the thickness grade for the appropriate material category and thickness range in Table 1 in accordance with the requirements of 6.1, except that for renovation using zinc paints, the thickness of renovation shall be 50 % higher than that required by Table 1, but not greater than 4.0 mils.
6.2.4 When areas requiring renovation exceed the criteria previously provided, or are inaccessible for repair, the coating shall be rejected."
I hope this answer helps. Thanks for your question.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
October 2015
Q: "What should be the weight of zinc for maximum protection of tubes in sea water environment?"
Answer:
The weight of the zinc on any article after galvanizing depends on the thickness of the article being galvanized. Normally, galvanizing will add 4% to 8% to the nominal weight of a steel article. Heavier items normally pick up a lower percentage of weight (4%-5%) than thinner material. Lighter, thinner materials pick up a proportionally heavier percentage, and usually show a 7% to 8% increase.
Here’s why. Hot dip galvanizing forms a metallurgical bond between steel article and the zinc in the galvanizing kettle. The coating “grows” from the surface of the steel, and the rate of growth slows as the steel reaches the temperature of the galvanizing bath. Thin materials heat rapidly, and thicker materials take longer to heat and develop thicker coatings. The specifications for galvanizing are covered by ASTM Iron A123 Standard Specification for Zinc (Hot-Dip Galvanized) Coatings on and Steel Products.
The following tables reflect the minimum average coating thicknesses required by ASTM A123.
The tables show that a quarter-inch (1/4") thick pipe or tube will develop a theoretical minimum of 3.0 mils on any given surface, and thinner materials will be proportionally lighter in coating. These are Minimum Average Coating thicknesses. Your galvanizer is required to meet these standards in every case, but due to the reactivity of certain steels it is not unusual for the galvanizer to exceed the minimum and provide a slightly heavier (thicker) zinc coating.
Since zinc service life is proportional to coating thickness, heavier coatings last longer than thinner coatings in the same given environment.
I hope this answer helps. Thanks for your question.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
August 2015
Q: "What thickness should the base material (steel) be to create 1/4" thick galvanized steel? What spec is the call out for the galvanized process?"
Answer:
Hot dip galvanizing forms a metallurgical bond between steel article and the zinc in the galvanizing kettle. The coating "grows" from the surface of the steel, and the rate of growth slows as the steel reaches the temperature of the galvanizing bath. Thin materials heat rapidly, and thicker materials take longer to heat and develop thicker coatings. The specifications for galvanizing are covered by ASTM Iron A123 Standard Specification for Zinc (Hot-Dip Galvanized) Coatings on and Steel Products. This specification covers the requirements for zinc coating (galvanizing) by the hot-dip process on iron and steel products made from rolled pressed and forged shapes, castings, plates, bars, and strips, or ASTM A153 Standard Specification for Zinc Coating (Hot-Dip) on Iron and Steel Hardware. This specification is intended to be applicable to hardware items that are centrifuged or otherwise handled to remove excess galvanizing bath metal (free zinc). Coating thickness grade requirements reflect this.
The following tables reflect the minimum average coating thicknesses required by ASTM A123.


As you can see, a 1/4" thick structural shape will develop a minimum coating thickness of 3.9 mils on any given surface. I should point out that if you are dealing with critical dimensions, ASTM A385 Standard Practice for Providing High-Quality Zinc Coatings (Hot-Dip) states, "When an assembly of steel parts or an individual steel part has been designed and fabricated with critical dimensions and must be subsequently hot-dip galvanized, the designer should be aware that the hot-dip galvanizing process uses chemical cleaning to prepare the surfaces for coating and this may alter the critical dimensions." In addition the hot-dip galvanized coating can increase the base steel material thickness from 2.0 to over 10.0 thousandths of an inch [50 to over 250 micrometres] depending on the steel chemistry as described in Section 3. This should be accounted for in the critical dimension design. Re-machining or otherwise reprocessing of critical dimensions after hot-dip galvanizing may be required.
I hope this answer helps. Thanks for your question.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
June 2015
Q: "We do stamping parts of various sizes but mostly small for the electrical industry. Parts are 3" to 24" long. We are facing issues of black spots and lumps on the parts during galvanizing. What can we do to correct this?"
Answer:
Thanks for sending pictures of the problem, and your competitors product. They make it easier to identify the issue and to offer some suggestions.
Some of the parts shown were clearly galvanized on a hook or wire using the existing hole in the flat bar with the bottom of the hook as the drain end of the article. The accumulation of zinc “lumps” is at the lowest point of the piece, and is where the zinc chills during withdrawal from the molten bath metal.
Another photo shows weld defects that are beyond of the control of the galvanizer. There is weld spatter, weld wire and undercut weld where the hook is welded to the flat. The right side of the photo shows zinc accumulation from drainage as described above
However, the parts formed from flats do not appear to outside of the norm for galvanized parts.
Please allow me to expand on the limits of the specifications for hot dip galvanizing; In general, galvanized coatings are specified because of their corrosion resistance, not because of their appearance. The coating standards are written on the basis MINIMUM coating weight (mass), and do not include a maximum, since coating growth is also determined by the chemistry of the steel being galvanized.
The galvanizing specifications note that finish is a subjective term and may be different from reader to reader. The spec states, “Finish—The coating shall be continuous (except as provided below), and as reasonably smooth and uniform in thickness as the weight, size, shape of the item, and necessary handling of the item during the dipping and draining operations at the galvanizing kettle will permit. Except for local excess coating thickness which would interfere with the use of the product, or make it dangerous to handle (edge tears or spikes), rejection for non-uniform coating shall be made only for plainly visible excess coating not related to design factors such as holes, joints, or special drainage problems. Since surface smoothness is a relative term, minor roughness that does not interfere with the intended use of the product, or roughness that is related to the as-received (un-galvanized) surface condition, steel chemistry, or steel reactivity to zinc shall not be grounds for rejection.”
If you desire a product that is cosmetically perfect there are several options that you may consider.
1. Processing material in smaller batch sizes. This allows the parts to be handled in such a way as to allow for sweeping of the parts during withdrawal and to minimize drips.
2. Use a mechanical or pneumatic vibrator to vigorously shake the parts during withdrawal from the galvanizing bath. This process will normally eliminate or minimize the formation of drips.
3. Alloy the galvanizing bath using Bismuth to increase fluidity of the metal and to promote better drainage.
4. Galvanize the parts in a centrifugal process to see if it is able to remove the excess zinc from the part and provide a better finish. ** CAUTION: Centrifugal galvanizing may remove too much zinc, so that the part does not comply with your minimum specified thickness.**
In summation, your competitor’s part does appear smoother, but is it a single example versus a complete batch of parts? Does the minor roughness at the base and the drip on the hook interfere with the intended use of the part, or is it purely cosmetic in nature? If it does not interfere, it is not grounds for rejection, unless it is by prior agreement between the contracting parties.
You may wish to download the app titled “Inspection of Hot Dip Galvanized Steel” for additional information. The app is free and is available for both Apple and Android devices.
Thank you for your question. I hope this answer helps.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
April 2015
Q: "Can 1/4'' QT100 plasma cut web plates on a trailer frame rail be galvanized without causing problems with cracking, etc. on the cut out sections of the plates?"
Answer:
QT100 is a quenched and tempered steel with excellent abrasion-resistant qualities, but the material isn't really well suited for the galvanizing process. Here's why.
First, the manganese content is outside the recommended parameters for achieving a normal coating. The recommended max for manganese is less that 1.3%, and QT100 is 1.5%. The result is a coating that doesn't develop normal thickness. The coating may be heavier and more subject to handling damage.
Most important is the Rockwell C of the QT material. Your plate has a RC of 52-53, which translates to a tensile equivalent of @ 265 ksi, which is far in excess of recommendations for galvanizing. The material will be brittle and cracking is very likely as a result.
In short, I would avoid the QT100 material.
Thanks for your question. I hope this helps.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
February 2015
Q: "To avoid losing small parts we would like to wire them to the parent assembly. Is this an acceptable practice?
What type and gage wire should be used?"
Answer:
It is completely acceptable to wire small parts to a parent assembly. Clip angles, match drilled plates, and other small detail parts are routinely wired to their larger components before galvanizing to insure that they are returned with the same piece.
The parts should be wired with not less than No. 9 gauge wire (0.148 in [3.76mm]). They should be wired so that they hang freely and don’t come into contact with the parent part in such a way as to cause a bare area or stick together during galvanizing.
The parts should be wired loosely through a hole so that the wire can be easily cut for removal of the part upon their return to you. (Don’t twist the wire so tight it sticks when galvanized.)
Contact your local galvanizer for their specific requirements, and be sure to note on any purchase documents that the component parts should not be removed from their parent assembly before galvanizing.
I hope this helps. Thank you for your question.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
December 2014
Q: "Is it possible to take a pipe spool which was recently galvanized, and re-galvanize it without stripping the old galvanizing off? The spool has bare spots from welding changes."
Answer:
The short answer is “No”. Welding through galvanizing has removed the coating at the weld area and in the surrounding heat affected zone of the weld. Zinc melts at 786 degrees C and during welding produces a voluminous white zinc oxide at about 1000 degrees F. Welding temperatures are in the range of 3-4000 degrees F or higher, depending upon the method.
The hot dip galvanized coating is formed through a diffusion reaction between iron and zinc and develops a metallurgical bond between the two metals. In order for the iron-zinc alloy to develop the steel substrate must be completely clean. Residual weld fluxes, oils, paint or any other contaminant will result in a bare area that will not galvanize. Attempting to re-galvanize without removing the existing zinc will result in a coating with bare areas and zinc that is not adherent to the pre-existing coating.
Galvanized steel develops zinc oxide on the surface of the material within 24 hours of the galvanizing process. The coating gradually weathers to form zinc hydroxide and finally zinc carbonate. The re-galvanizing process will require that any zinc on the steel surface be removed completely before being re-coated in order to meet ASTM specifications.
ASTM allows for repair, but limits the allowable area. The total area subject to renovation on each article shall be no more than 1⁄2 of 1 percent of the accessible surface area to be coated on that article, or 36 in.2 per short ton [256 cm2 per metric ton] of piece weight, whichever is less. The specification further state, “Materials that have been rejected for reasons other than embrittlement are not prohibited from being stripped and re-galvanized and again submitted for inspection and test at which time they shall conform to the requirements of this specification.”
I hope this helps. Thank you for your question.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
October 2014
Q: "For a mild steel structure which is going to support solar panels in outdoor applications where climate conditions will vary from dry to high humidity, is electroplating or hot dip galvanizing the better choice?"
Answer:
Hot dip galvanizing is the better choice. Electroplating is a process in which zinc is electro deposited onto the steel surface from a zinc salt solution. The coatings are typically very uniform, but very thin, ranging up to just 1 mil (25 µm), restricting zinc-plated parts to very mild (indoor) exposures.
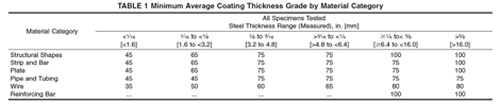
ASTM Specification B 633 lists four classes of zinc plating: Fe/Zn 5, Fe/Zn 8, Fe/Zn 12 and Fe/Zn 25. The number indicates the coating thickness in microns (µm). Hot dip galvanizing provides the heaviest commercial available zinc coating. Steel is chemically cleaned and then immersed in a bath of molten zinc metal. The coating forms a metallurgical bond between zinc and steel that is superior to the bond of plating. The coating thickness for hot dip galvanizing will be dependent upon the thickness of the base steel being galvanized. See Table 1 for the list of coating thicknesses by category.
It is important to note that the service life of a zinc coating is dependent upon the thickness of the coating. A coating that is 25 µm thick will last only half as long as a coating that is 50 µm in the same exposure conditions.
The best choice for your solar application is hot dip galvanizing.
Thanks for your question.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
August 2014
Q: "How can I calculate the percentage of zinc on my galvanized parts? I want to confirm that they do in fact contain 98% zinc in the galvanizing."
Gloria Reyes
Answer:
The specifications for zinc are defined by ASTM B6, Standard Specification for Zinc. Galvanizers in North America use one of three types of zinc in their galvanizing bath.
1. Prime Western – 98.5% Zinc
2. High Grade – 99.5% Zinc
3. Special High Grade – 99.95% Zinc
Imported galvanized material from EU nations will be very similar to North American galvanized products in composition and will have the same zinc levels.
If you are importing galvanized articles from China you are open to materials that do not conform to ASTM specifications and zinc metal that may contain a variety of elements that you may not be interested in owning.
You could simply ask your galvanizer for a copy of their bath metal analysis. Most galvanizers analyze bath chemistry frequently to insure that they keep their alloying elements within a specific range, and most will be happy to provide this information to their customers at no cost.
Or, you could hire a laboratory consultant, send a sample of the material for x-ray diffraction analysis or use a gas chromatography–mass spectrometry, spend big bucks, and get the same answer.
Hope this helps. Thank you for your question.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
June 2014
Q: "Is there such a thing as Marine Grade Galvanizing and if so how does it differ to standard galvanizing?"
Jeff Holmes, Coffs Harbour City Council,
NSW, Australia
Answer:
This is a new one on me, and is likely being used as a marketing gimmick to promote the products of a specific vendor. Some US manufacturers use the term “marine grade” for hot dip galvanized wood screws. This is to differentiate themselves from electroplated or mechanically plated fasteners that have a much thinner coating. Hot dip galvanizing is the heaviest commercially available method for applying a protective zinc coating to a steel product, and as such it will be better and for a longer time period than any other type of zinc coating. Zinc performance is linear. Thicker coatings result in longer life. Period.
The hot dip galvanized coating is formed through a diffusion reaction during immersion in the molten zinc and it develops a metallurgical bond between the zinc coating and the steel substrate. When the coating is fully developed, the reaction between the two metals ceases and the steel is removed from the bath. Subsequent immersion in the zinc bath may result in (1) a marginal increase in coating thickness or (2) a heavier coating that is brittle and subject to flaking.
Simply specifying a coating to AS/NZS 4680 specification will ensure that you get the best possible galvanized coating for the article being coated. Purchasing “marine grade” may sound better and will probably cost more, but will not be different from any other hot dip coating.
I hope this helps, and thank you for your question.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
April 2014
Q: "We are a steel poles manufacturing company. Some of our customers are asking for a high coating thickness of 120 microns for low silicon material. We are taking more dipping time to achieve this, but we are getting additional rough coating accumulating in the seam welding line. How can we avoid these additional microns in the welding line?"
Answer:
ISO 1461 and ASTM A123 both require a 100 µm nominal coating thickness for steel with a base thickness of ¼” or more (≥6.4 mm). The additional 20 microns that your customer requires will require additional kettle time, especially if the steel is low silicon in composition.
In order to achieve the same coating thickness, it is recommended that the weld filler metal be comparable chemistry to the steel of the base material. If the weld filler metal is a different composition than the steel being joined, it may react with the zinc at a different rate and give a thicker, or thinner, coating over the weld. To prevent an increased reaction of the weld material with the molten zinc, the filler material should have less than 0.3 percent silicon content.
For typical welding processes, such as shielded metal arc welding (SMAW), submerged arc welding (SAW) and flux-cored arc welding (FCAW), there are weld rod materials that will not cause excessively thick coatings. The table below indicates the material and chemistry for several welding rods that yield good coating appearance and thickness.
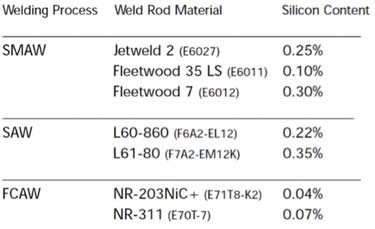
I hope this answer helps. Thank you for your question.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
December 2013
Q: "What is the additional weight per square foot of surface area of galvanizing on a steel member?"
Answer:
Good question. Normally, galvanizing will add 4% to 8% to the nominal weight of a steel member, depending upon the thickness of the steel being galvanized. Galvanizing is an alloy that is formed through a diffusion reaction between iron and zinc. The steel is allowed to remain in the galvanizing bath until the steel core reaches the temperature of the surrounding zinc bath, when the galvanizing reaction is complete. Thicker (heavier) steels take longer to heat, so the reaction continues for a longer period. Thinner (lighter) steels absorb heat more quickly and complete their reaction sooner and develop a lighter coating mass.
It seems counter intuitive, but heavier steels normally pick up a lower percentage of weight (4%-5%) due to the weight/surface area ratio, than thinner material. Light sections pick up a proportionally heavier percentage due to the same effect, and usually show a 7% to 8% increase in mass.
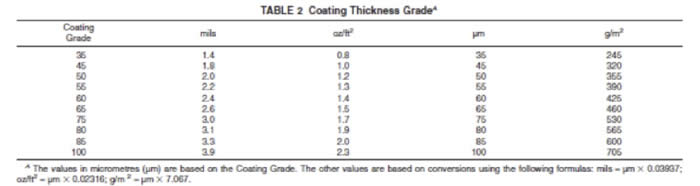
The ASTM Standards provide for MINIMUM coating weights according to the tables below:

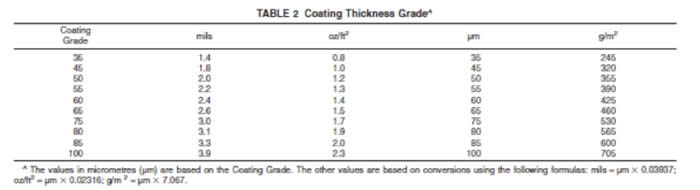
Please remember that these are MINIMUM coating thickness standards. Galvanizing will normally meet or exceed these minimums in every case. A steel beam that has a nominal thickness of >1/4” will result in a galvanized coating of 100 micrometers, that will add a minimum of 2.3 oz/ft2 to the weight of the article.
Thank you for the question!
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
October 2013
Q: "In the gas piping in Hong Kong, galvanization is applied to both low carbon steel pipe (EN10255) and malleable iron fittings (ASTM A197). Are the metallugical bonds between zinc and iron substrate different in these cases? Our experience has been that the galvanized iron fittings are more corrosion resistant than galvanized steel pipe. Can this be explained by the graphite content of malleable iron?"
Answer:
The metallurgical bond between iron and zinc will be the same for any ferrous material. I would suspect the differing corrosion rates you are experiencing are not due to the graphite content, but rather from the different amount of zinc that is deposited on the two different materials.
It is known that the exact structural nature of the galvanized coating may be modified in accordance with the exact chemical nature of the steel being galvanized. Certain elements found in steels are known to have an influence on the coating structure. The elements carbon in excess of about 0.25 %, phosphorus in excess of 0.04 %, or manganese in excess of about 1.3 % will cause the production of coatings different from the “normal” coating.
Steels with silicon in the range 0.04 % to 0.15 % or above 0.22 % can produce galvanized coating growth rates much higher than those for steels with silicon levels below 0.04 % and between 0.15 % and 0.22 %. Recent studies have shown that even in cases where the silicon and phosphorous are individually held to desirable limits, a combined effect between them can produce a coating which typically would have a mottled or dull gray appearance.
These more reactive coatings are generally heavier (thicker) than normal. The corrosion rate of zinc is linear. Two mils of zinc will have exactly 2X the service life of one mil of zinc under identical exposure conditions. Consequently, thicker coatings have a longer life. Additionally, galvanized pipe is usually “wiped” during withdrawal from the bath using air or steam “knives” to remove excess zinc from the surface and provides a very smooth, uniform appearance for the consumer.
The relative coating thickness of the two materials can be non-destructively tested using a magnetic thickness gauge. This will quickly tell if the coating meets your minimum requirements.
Thank you for the question!
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
August 2013
Q: "Who's responsible for taking the mill markings off of channel before it's galvanized, the fabricator or the galvanizer?"
Answer:
The fabricator. Galvanizing is specified to ASTM A123 Standard Specification for Zinc (Hot-Dip Galvanized) Coatings on Iron and Steel Products for channel shapes. The specification contains “Referenced Documents” in the body of the standard and includes ASTM A385 Practice for Providing High-Quality Zinc Coatings (Hot-Dip). A385 is often referred to as the “design spec”. A385 includes the following in Section 14, Marking for Identification. 14.1 Paint is not removed by pickling and must not be used when marking for identification material to be galvanized. Mill markings are sometimes referred to as "ink", but the composition is really that of paint.
Many fabricators mistakenly assume that the acid (pickling solution) used by the galvanizer will remove paint, and this is incorrect. In fact, some paint formulations are used as a “masking” for areas that will require full-penetration welding after the galvanizing process, such as welded moment connections. During galvanizing the paint burns and converts to surface carbon contaminant which is easily removed, exposing the bare steel substrate, ensuring good weld penetration.
If the mill markings remain on the steel surface before galvanizing they are clearly visible after galvanizing. The appearance is similar to a bleed-through marker used for paint coatings.
Some mill markings are soluble in the caustic and acid and are easily removed, while some mills use a formulation that requires grinding by the fabricator before being sent to a galvanizer.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
June 2013
Q: "What's the inspection process for galvanizing a large fabricated steel truss? Not all the elements are the same thickness (base plate 10mm, frame 4mm). Won't the smaller, thinner parts throw off the average coating thickness?"
Answer:
Materials of different thicknesses must be treated separately for the purpose of inspection. If there are two basic parts to the weldment – base plate and frame, for example – you'd need an acceptable average coating on each of them for it to pass inspection, not one overall average. Different thicknesses of plate and different thicknesses of frame will also need to be measured independently. Each type and thickness of material has its own requirements.
Structural steel fabrications are galvanized to ASTM A123, Standard Specification for Zinc (Hot-Dip Galvanized) Coatings on Iron and Steel Products.
The coating thickness requirements for galvanizing are determined by the thickness of the base metal being galvanized. The galvanized coating is formed through a diffusion reaction between iron and steel. As the steel core approaches the temperature of the galvanizing bath the rate of diffusion of the coating slows, and finally stops. Thinner materials will reach their theoretical maximum coating faster than thicker steel, which take longer to heat. Therefore, different thickness material in the same weldment will have different coating thickness, as shown in the tables below. The first table shows the coating grade required for different materials at different thicknesses. The second table indicates the coating thickness for each grade.
All Specimens Tested Steel Thickness Range (Measured), in. (mm)
Material Category _____________________________________________________________________
<1/16 1/16 to <1/18 1/8 to 3/16 >3/16 to <1/4 ≥1/4
(<1.6) (1.6 to < 3.2) (3.2 to 4.8) (>4.8 to 6.4) (≥6.4)
Structural Shape and Plate 45 65 75 85 100
Strip and Bar 45 65 75 85 100
Pipe and Tubing 45 45 75 75 75
Wire 35 45 75 75 75
Reinforcing Bar ---- ---- ---- ---- 100
---------------------------------------------------------------------------------------------------
Coating Grade Thickness (mils)
35 1.4
45 1.8
50 2.0
55 2.2
60 2.4
65 2.6
75 3.0
80 3.1
85 3.3
100 3.9
Our colleague, Dr. Galv at the AGA, received a similar question a while back. If you'd like to read his answer, click here.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
April 2013
Q: "I have found patches of what looks like thick white rust on galvanized steel bars. Can they still be used? Does this threaten the integrity of the coating?"
Answer:
What you are describing is 'wet storage stain.' It's the result of recently galvanized steel being stored or transported in wet or poorly ventilated conditions. Whether or not it is a threat to the coating depends on the severity of the problem, but it most cases, it is not. Because it is voluminous, wet storage stain usually looks worse than it is. In the vast majority of cases, wet storage stain does not indicate serious degradation of the zinc coating, nor does it necessarily imply any reduction in the expected life of the product.
Wet storage stain is a thick white or gray deposit formed by accelerated corrosion of a new zinc coating in wet or humid conditions. It is found most often on stacked and bundled materials, such as galvanized sheets, plates, angles, bars and pipe, where tight packing can trap moisture. A thin film of water caught between two pieces of fresh zinc coated steel creates an environment where exposure to oxygen and carbon dioxide are limited, preventing the formation of a protective zinc carbonate layer and allowing the growth of soft corrosion products.
While very unsightly, even medium or heavy layers of wet storage stain represent the loss of very little zinc from the base coating. Light areas of wet storage stain, such as those you described, will usually be converted to zinc carbonate and will disappear in use with exposure to normal atmospheric conditions.
Medium to heavy wet storage stain should be removed as much as possible before the steel is put into service. The American Galvanizers Association recommends cleaning wet storage stain of "medium severity" a mild solution of ammonia (one part ammonia to 10 parts fresh water) and a nylon bristle brush. For heavier wet storage stain, a mild acidic solution having a pH of 3.5 to 4.5, such as acetic acid or citric acid mixed with fresh water, and a nylon bristle brush can be used. Get more details here.
This is not to say that wet storage stain is never a serious problem. It can be. When wet storage stain is very heavy, as indicated by black spots on the galvanized coating or rusting of the underlying steel, the integrity of the coating is certainly damaged. At that point, stripping and re-dipping may be necessary.
One last note from the AGA: "While the galvanizer has a responsibility to produce a galvanized coating of good quality in conformance with governing specifications, the galvanizer is no longer in control after the work leaves the plant. The purchaser must ensure the proper handling and storage of material at the construction site to ensure a stain-free product at the time of final installation." See "Prevention of Wet Storage Stain" on page 3 of the attached article.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
February 2013
Q: "I have been asked to obtain galvanized shim 150mm x 150mm x 2mm and 5mm (150 pcs of each) 10mm and 20mm (80 pcs of each). Would this be a possibility? My usual supplier advised not, as each item would need a hole to enable dipping. Do you think I could find someone that would be able to supply them without a hole?"
Answer:
If you look hard enough, I believe that you will be able to find someone who can do this without adding a hole. But, I believe you will to pay a significant premium for this service.
Hot dip galvanizing takes place at @ 450 C. The addition of ambient temperature steel to a very hot zinc bath results in a volatile mix. Zinc is generally thrown from the kettle surface during the immersion of steel, and light weight parts may be thrown from their fixture or rack during this phase.
Zinc and steel have nearly the same specific gravity. Zinc is about 7.12 and steel is about 7.82, so the steel actually becomes buoyant in the molten zinc. Once again this may result in parts "floating" from their fixture and loss of parts in the galvanizing kettle. Some of the shims you describe weigh less than .35 Kg to more than 3.5 Kg.
The flat geometry of a shim doesn't facilitate centrifugal galvanizing. The parts lie together and freeze into a solid block of shims as the zinc cools during withdrawal from the galvanizing bath after a spinning operation. If no handling hole is provided they would require a special rack or fixture to keep them from being lost in the bath, and to achieve a quality finish without the parts sticking together.
If the shims have a 1 cm hole added to one corner they can be wired in such a way that they can be dipped without loss of the part and achieve a finish that will be suitable for use with very little finishing or filing to remove drips or runs. This is certainly the most economical method of handling and will likely give you a lower cost on the part.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
December 2012
Q: "What is the difference between galvanized and triple zinc galvanized?”
Answer:
Imagination! Triple zinc galvanized is a marketing gimmick to convince you, the user, that you’re getting a galvanized coating that has three times the life of conventional galvanized steel. I generally describe this type of sales promotion as “ferro-manurium”. (Another imaginary corrosion terminology.)
One manufacturer describes his Triple Zinc (TZ) galvanizing as a prefabrication coating of 1.85 (G-185) ounces of zinc per square foot of surface area measured in accordance with ASTM A 653 Standard Specification for Steel Sheet, Zinc-Coated (Galvanized) or Zinc-Iron Alloy- Coated (Galvannealed) by the Hot-Dip Process. G-185 is a measure of the total coating weight expressed in ounces of zinc per square foot of surface area.
Keep in mind that the single side measurement will be half of the total and due to variables in the process there may be as much as a 60/40 distribution difference between the two sides. A G-185 coating would have roughly 1.5 mils of galvanized coating on any given surface. A normal commercial grade A-653 coating is galvanized to G-60, so a G-185 would be three times the amount of zinc.
A tubing manufacturer uses the terminology for a continuous in-line hot dip galvanizing process, then a conversion process (probably chromate passivation) then a clear organic topcoat.
In comparison, a hot dip galvanized coating to ASTM A123 Standard Specification for Zinc (Hot-Dip Galvanized) Coatings on Iron and Steel Products will have a coating thickness that will vary depending upon the thickness of the steel substrate. The thinnest coating on steels with a thickness <1/16” will have a minimum coating thickness of 1.8 mils and heavier material ≥1/4” will have a minimum of 3.9 mils of zinc. These coatings are based on single side measurements, so the total coating weight will be twice the thickness of a single measurement. Even the thinnest A123 coating will exceed the coating thickness of the triple zinc G-185 coating. This is important since the corrosion rate of zinc is linear. Two mils of zinc will last twice as long as one mil of zinc in the same application. (More is better.)
I hope this helps answer your question.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
October 2012
Q: "Does ASTM mandate inspection after galvanizing as part of meeting the spec?”
Answer:
A. Absolutely. For the purposes of this discussion I will limit my remarks to ASTM A123, Standard Specification for Zinc (Hot Dip Galvanized) Coatings on Iron and Steel Products.
The specification defines inspection in both inch-pound and SI (metric) units of measure. The galvanizing standards are essentially based upon minimum coating thickness measurements for articles of a defined surface area. The number of measurements is defined by the specimen size and a random statistical sampling of an entire lot of parts.
Section 9 of the standard, Inspection, Rejection and Retest, states, “It is the responsibility of the galvanizer to ensure compliance with this specification. This shall be achieved by an in-plant inspection program designed to maintain the thickness, finish, and appearance within the requirements of this specification unless the inspection is performed in accordance with 9.2 Inspection by the Purchaser.
Section 10 states, “When specified in the purchase order or contract, the purchaser shall be furnished certification that samples representing each lot have either been tested or inspected as directed by this specification and the requirements have been met. When specified in the purchase order or contract, a report of the test results shall be furnished."
All of this being said it is important to remember that A123 is a coating thickness standard. There will be variation in color of the galvanized coating that the galvanizer has no control over. Steel chemistry of the article being coated effects the final appearance of the galvanized surface. Smoothness is another variable that is often questioned, since smoothness is a relative term and highly subjective in nature. Roughness that does not interfere with the intended use of the article shall not be grounds for rejection.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
August 2012
Q: "Is the substrate for sheet coil HDG cold rolled or hot rolled?”
Answer:
A. Either is acceptable. ASTM A653 Standard Specification for Steel Sheet, Zinc-Coated (Galvanized) or Zinc-Iron Alloy-Coated (Galvannealed) by the Hot-Dip Process is the spec relating to the actual coating of the substrate, but also references A568/A568M Specification for Steel, Sheet, Carbon, Structural, and High-Strength, Low-Alloy, Hot-Rolled and Cold-Rolled, General Requirements for, as the description for the substrate being galvanized.
During the manufacture of steel if the temperature of the metal is above its recrystallization temperature, or the temperature at which the grain structure of the metal can be altered, then the process is termed as hot rolling. If the temperature of the metal is below its recrystallization temperature, the process is termed as cold rolling.
In hot rolling, the process removes any induced stresses and grain deformation due to the elevated working temperature. During the cooling process, non-uniformed cooling may occur, which results in residual stress of the product. Hot rolling is used mainly to produce products like sheet metal or simple cross sections, such as rail tracks and I-beams.
Cold rolling has the added effect of work hardening and strengthening the material thus further improving the material’s mechanical properties. It also improves the surface finish and holds tighter tolerances. However, room temperature steel is less malleable than hot steel, so cold rolling cannot reduce the thickness of a work piece as much as hot rolling in a single pass. Commonly cold-rolled products often include similar hot rolled products like sheets and bars, but are usually smaller. Cold rolled material is generally more expensive than hot rolled due to the additional time and costs related to the process.
The substrate can be either hot or cold rolled, but due to the tolerance requirements of most coil products, it is very likely that you are going to get cold rolled material when you specify A653.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
June 2012
Q: "I have 12 gauge steel plate with 1/4" diameter holes. How should I spec this material out to be galvanized without having the holes fill with zinc?”
Answer:
A. 12 gauge is actually referred to as “sheet”. The difference is a matter of thickness. For example steel PLATE is a large plate of thickness greater than 3/16 inch (about 5 mm). Steel SHEET is plate that it LESS than 3/16 inch thick. And steel STRIP is SHEET cut into short widths. 12 gauge is approximately .1094 inch (2.8 mm) in thickness. If this sheet is galvanized to ASTM A123 Standard Specification for Zinc (Hot Dip Galvanized) Coatings on Iron and Steel Products, the spec requires that the MINIMUM average coating thickness requirement is 65μm (2.6 mil) for material that is 1/16” to < 1/8” (1.6 to 3.2 mm).
Section 6.4 Appearance, contains the notation that plain holes of ½-in (12.5 mm) or more shall be clean and reasonably free from excess zinc. The section also contains a note that states “Depending upon product design or material thickness, or both, filming or excess zinc buildup in plain holes of less than ½-in (12.5mm) diameter may occur that requires additional work to make the holes usable as intended.”
ASTM A385 Standard Practice for Providing High-Quality Zinc Coatings (Hot-Dip) also referred to as the “design spec” makes note that for venting purposes holes shall not be less that 3/8” (9.5 mm) in diameter.
Hot dip galvanizing requires dipping of the chemically cleaned steel part in a bath of molten zinc at about 840 F, and allowing the part to reach the temperature of the bath to complete the metallurgical bond of iron and zinc. Thinner materials heat rapidly, and thicker materials take longer in the bath to galvanize. The rate of cooling also varies with thickness. Thicker material will stay hotter longer, and allow the excess zinc to drain more effectively. Thinner materials cool rapidly, and will capture more zinc as the temperature drops to 784 F where zinc changes from liquid to solid phase.
¼” diameter holes are too small to galvanize without the holes filling or filming with zinc. Fortunately however, the pure zinc surface is relatively soft (70 DPN) and can be easily removed by chasing or reaming the hole after galvanizing. Alternatively, you may want to consider using pre-galvanized ASTM A653 sheet stock and simply punching the holes. The zinc surface will provide cathodic protection to prevent corrosion at the edge of the punched hole.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
April 2012
Q: "Do extreme temperatures affect the corrosion resistance of galvanized steel?”
Answer:
A. "Extreme" is a subjective term. The possible range of normal outdoor environmental air temperatures has little effect hot-dip galvanizing's corossion resistant properties.
In fact, according to the AGA, "hot-dip galvanized steel does not show significant differences at very low temperatures, below -40 F, or at very high temperatures, above 150 F."
Galvanized steel is a very good choice for low temperature environments. Studies done on the effect of low temperature environments on hot-dip galvanized steel indicate only minimal change in the behavior of the zinc coating. Some polar installations have used galvanized steel for corrosion protection and have been in service for many years. As with any steel at extremely low temperatures, the material may become brittle with extended use.
Very high temperatures can have an effect on galvanized steel depending on the length of exposure and the severity of the environment. The recommended short-term maximum temperature exposure for galvanized steel is around 660 F for periods of less than two hours at a time, or for a one-time occurrence lasting less than 24 hours.
When considering long-term exposure, the recommended maximum service temperature is approximately 390 F (200 C). Problems that could occur from long-term exposure to temperatures above 390 F include peeling, which can cause the outer free zinc layer to separate and fall away from the underlying zinc-iron alloy layers. It should be noted, however, the remaining zinc-iron alloy layers can still provide a high level of corrosion protection at temperatures below 480 F (250 C).
Extended exposure to high temperatures above 480 F will accelerate peeling and can result in the zinc-iron alloy layers cracking and separating from the steel. That's pretty extreme. Hot dip galvanizing protects steel from corrosion "extremely" well in the high and low temperature ranges of most applications.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
December 2011
Q: "What care is taken for galvanizing threaded parts, and is rethreading them allowed after galvanizing?”
Answer:
A. Fasteners and other threaded parts are galvanized to ASTM A153/A123M, Standard Specification for Zinc Coating (Hot-Dip) on Iron and Steel Hardware. This specification is intended to be applicable to hardware items that are centrifuged or otherwise handled to remove excess galvanizing bath metal from the parts.
Bolts and similar items are galvanized in perforated metal baskets and the parts are centrifuged at a high rate of speed during withdrawal from the galvanizing bath to remove excess metal from the threads. Nuts are galvanized as “blanks” with no internal threads, and the threads are cut into the nut after galvanizing. The threads on the nuts are cut slightly oversized to allow for the buildup of zinc on the surface of the male thread on the bolt. The amount of overtap is dependent upon the thickness of the coating, which is in turn dependent upon the thickness of the steel article. (Thicker materials = thicker coating)
There will be no corrosion on the bare threads of the nut, because the steel surface is in direct contact with the galvanized coating on the bolt. The zinc on the bolt will protect the bare threads on the nut through its anodic reaction.
Section 4.4 of the specification Threaded Articles—states “the zinc coating on threads shall not be subjected to a cutting, rolling, or finishing-tool operation, unless specifically authorized by the purchaser. In order to meet overtapping allowances, tapping the threads of nuts or tapped holes after galvanizing is not prohibited."
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
October 2011
Q: "At what depth should someone engrave a steel plate so that, when galvanized, it can still be read? What is the minimum depth and the best depth?”
Answer:
Thanks for the question. It is a fairly common requirement that marking be legible after galvanizing for most fabricated steel structures, and it is covered by ASTM A385, Standard Practice for Providing High Quality Zinc Coatings (Hot Dip). Section 14.2 Marking for Identification states, “Satisfactory identification may be provided by welding the identifying marks on the material, by embossing the identifying marks on a steel tag of no less than No. 12 gage (0.105 in. [2.69 mm]) and securing to the material with a heavy wire such as No. 9 gage (0.148 in. [3.76 mm]), or by die stamping the identifying marks into the material with characters 1⁄2 in. [12.7 mm] high and a minimum of 1⁄32 in. [0.79 mm] deep.
The galvanized coating has a minimum thickness of 4 mils (0.004”) for material that is ¼” thick. There will be some variation in the measurement over the surface of an article, so for engraving, deeper is better to prevent the cut from filling with zinc.
Please let me know if you have any additional questions or if I can be of additional assistance.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
August 2011
Q: "What does galvanizing add to the thickness of steel?”
Answer:
Galvanizing is an alloy that forms when the prepared steel surface is immersed in a bath of molten zinc. The galvanizing bath temperature is @ 840-845 degrees F, and the steel must remain in the bath until the thickest part of the steel fabrication reaches the temperature of the bath. When the steel core reaches the bath temperature the reaction stops and the steel has been galvanized.
The coating reaction begins while the steel is heating and stops when it reaches bath temperature, so thin or light structures reach temperature faster than thick or heavy sections. Consequently the coating thickness will be heavier or thicker on the heavy material and the ASTM specifications reflect this difference in coating weight, based on the thickness of different materials.
There are other factors that can affect the coating, such as unusual steel chemistry, but I will keep the discussion for this based on standard mild carbon structural material.
Please refer to ASTM A123 for complete details on the minimum thickness values for different materials. The categories are Structural Shapes and Plates, Strip and Bar, Pipe and Tubing, Wire, and Reinforcing Bar. For the purposes of this question, I will discuss structural shapes and plates. The Steel Thickness Range (Measured), in. (mm) is as shown below in μm and mils.
<1⁄16 (<1.6) 1⁄16 to <1⁄8 (1.6 to <3.2) 1⁄8 to 3⁄16 (3.2 to 4.8) >3⁄16 to <1⁄4 (>4.8 to <6.4) ≥1/4 (≥6.4)
45 65 75 85 100
1.8 mils 2.6 mils 3.0 mils 3.3 mils 3.9 mils
The values in micrometres (μm) are based on the Coating Grade. The other values are based on conversions using the following formulas: mils = μm x 0.03937; oz/ft2 = μm x 0.02316; g/m 2 = μm x 7.067.
Based on this information, steels with a base metal thickness from 1/8” to ¼” will have a minimum coating thickness of 3 to 4 mils. (.003” to .004”) It is important to note that these are MINIMUM coating thicknesses. It won’t be unusual for the coating to exceed these minimums and be 4 to 6 mils in thickness.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
June 2011
Q: "Is there a way to predict the expected service life of galvanized steel in industrial use?”
Answer:
A. As you might expect, the answer depends on several factors, including the environment, the application and the thickness of coating.
The American Galvanizers Association (AGA) published the Time To First Maintenance Chart in 2010. ‘Time to first maintenance’ is defined as 5% rusting of the steel surface. According to the chart, in a typical industrial environment with the ASTM A 123 minimum 3.9 mil zinc coating, it will be 72 years before any maintenance is required. The coating thickness of hot-dip galvanized steel is usually between 4 and 5 mils. This means that the first need for surface maintenance may not occur for more than 90 years. Then you might need to do a little touch-up. The chart is available for download from the AGA Web site, http://www.galvanizeit.org/aga/resources/aga-publications/.
The Time To First Maintenance Chart was developed using a corrosion prediction model called the Zinc Coating Life Predictor (ZCLP). The program performs calculations based on models developed using statistical methods, neural network technology and an extensive worldwide corrosion database.
You can use the ZCLP yourself. Just click here [http://www.galvinfo.com:8080/zclp/] and put in your specific parameters. Obviously, atmospheric levels of airborne salinity, precipitation, relative humidity, sulfur dioxide, and temperature influence actual corrosion rates in specific geographic locations.
Annual average temperatures, precipitation and humidity for your state or city can be found at www.currentresults.com. You may have to estimate airborne SO2 and salinity levels for your site, unless you have actual test results. The following information might help in your estimating. According to the European Community’s LIFECON project (2003), concentrations of airborne sulfur dioxide from 60 mg/m2/day and up are considered Industrial, with 10 to 80 mg/m2/day being Light Industrial, and below 10 mg/m2/day being benign. Similarly, 60 or more mg/m2/day of airborne salinity is considered a Marine environment, with 15 to 60 mg/m2/day being Light Marine and below 15 mg/m2/day being benign. For coating thickness, since hot dip galvanizing is usually between 4 and 5 mils thick, I would go with 4.5. (Be sure to select ‘mils’ from the dropdown.)
Plug in the parameters that best describe your project and see what the ZCLP has to say. Please note that some of the parameters have drop-down selections. Make sure you choose the right ones. It will affect the results.
So, to answer your question, yes, there is a way to estimate the expected service life of galvanized steel in industrial applications. Keep in mind, the ZCLP’s predictions are estimates, not guarantees. Are they accurate? Most likely, but the fact is that hot dip galvanized steel lasts so long we may not be around to find out!
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
April 2011
Q: "In reference to G140, the minimum figure (for coating mass) is 1.40oz/ft2. Is there a nominal figure?” "
Answer:
G-140 is covered by the specification ASTM A653/A653M-09a, titled Standard Specification for Steel Sheet, Zinc-Coated (Galvanized) or Zinc-Iron Alloy-Coated (Galvannealed) by the Hot-Dip Process.
This specification covers steel sheet, zinc-coated (galvanized) or zinc-iron alloy-coated (galvannealed) by the hot dip process in coils and cut lengths. The product is produced in various zinc or zinc-iron alloy-coating weights [masses] or coating designations as shown in Table 1 of the standard.
Coating designation G140 does indeed indicate a coating mass of 1.40 oz/ft2 however, it is important to note that the measurement is for the total of both sides. Further, the 1.40 oz/ft2 is a triple spot test (TST) for the total of both sides. The coating thickness of a single side of G140 (TST) is approx 1.19 mils (1.4 oz/ft2 x 1.7 mils /2 = 1.19 mils)
The coating designation is the term by which the minimum triple spot, total both sides coating weight [mass] is specified. Because of the many variables and changing conditions that are characteristic of continuous hot-dip coating lines, the zinc or zinc-iron alloy coating is not always evenly divided between the two surfaces of a coated sheet; nor is it always evenly distributed from edge to edge. However, the minimum triple-spot average coating weight (mass) on any one side shall not be less than 40 % of the single-spot requirement.
The triple spot test (TST) for one side of G140 is 0.48 oz/ft2 and the single spot test as a total of both sides is 1.2 oz/ft2.
Use the following relationships to estimate the coating thickness from the coating weight [mass]:
1 oz/ft2 coating weight = 1.7 mils coating thickness,
7.14 g/m2 coating mass = 1 μm coating thickness.
Use the following relationship to convert coating weight to coating mass:
1 oz/ft2 coating weight = 305.15 g/m2 coating mass.
As it is an established fact that the atmospheric corrosion resistance of zinc or zinc-iron alloy-coated sheet products is a direct function of coating thickness (weight (mass)), the selection of thinner (lighter) coating designations will result in almost linearly reduced corrosion performance of the coating. For example, heavier galvanized coatings perform adequately in bold atmospheric exposure whereas the lighter coatings are often further coated with paint or a similar barrier coating for increased corrosion resistance. Because of this relationship, products carrying the statement “meets ASTM A653/A653M requirements” should also specify the particular coating designation.
Although the corrosion rate can vary considerably depending on the environmental factors, it is well known that, in most instances, the life of the zinc coating is a linear function of coating mass for any specific environment. That means, to achieve twice the life for any specific application, the user should order twice the coating mass.
Examples: A G60 coating mass will exhibit approximately twice the life of a G30 coating mass. A G90 coating mass will exhibit about 50% longer life than a G60 coating mass.
Products that are hot dip galvanized after fabrication are galvanized to ASTM A123, “Standard Specification for Zinc (Hot-Dip Galvanized) Coatings on Iron and Steel Products.”
This specification covers both unfabricated products and fabricated products, for example, assembled steel products, structural steel fabrications, large tubes already bent or welded before galvanizing, and wire work fabricated from uncoated steel wire. A123 specifications differ from continuous galvanizing specs in that the coating thickness or mass designation is not measured as a distribution between both sides of an article. If the specification calls for 2 oz/ft2 (3.4 mils) the coating will be as specified anywhere on the article. The total for both sides would be a minimum of 6.8 mils
I hope that this answers your question, and clarifies the mystery of “G” designations for sheet galvanized products.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
February 2011
Q: "What would be the best galvanized coating (i.e.G90, G140) for metal framing on an exterior wall of a high rise building on the oceanfront in Virginia Beach VA? What would be the life of your suggestion? Thank you in advance."
Answer:
Your question warrants a multi-part answer, which I will attempt to describe below.
G-90 or G-140 are both covered by ASTM A653 “Standard Specification for Steel Sheet, Zinc-Coated (Galvanized) or Zinc-Iron Alloy-Coated (Galvannealed) by the Hot-Dip Process”. This specification covers steel sheet, zinc-coated (galvanized) or zinc-iron alloy-coated (galvannealed) by the hot dip process in coils and cut lengths. These are mill galvanized materials and are limited to coil stock that is then cut to length and formed. This is the same material that HVAC duct work is formed from.
The weight of coating is specified by the “G” designation and describes the weight or mass of the coating as the Total Both Sides, oz/ft2 of zinc. G-90 is 0.9 oz/ft2 of zinc, and G-140 is 1.4 oz/ ft2 of zinc. But again this is the TOTAL of both sides of the galvanized article. So, a G-90 coating actually has only 0.45 oz/ ft2 of zinc on any given surface.
The accepted formula for coating mass is 1 oz/ ft2 of zinc = 1.7 mils of thickness. This means that a G-90 coating has 0.45 oz/ ft2 x 1.7 mils = .765 mils of thickness on any given surface. G-140 has 0.7 oz/ft2 x 1.7 mils = 1.19 mils of thickness on any given surface. (However since the coating distribution is not exact, the coating can actually be a 60/40 distribution from side to side.)
Zinc (galvanizing) service life is a linear measure, depending upon the type of environment and the thickness of the coating. Once you know the thickness of the zinc coating and the type of exposure, the service life of the coating is easily predictable.
Two mils of zinc will last exactly twice as long as one mil of zinc in the same exposure conditions. Therefore a G-140 coating will last about 36% longer than a G-90 coating for the same application.
For a more comprehensive comparison of sheet specifications and hot dip galvanizing after fabrications please click the following link;
http://www.galvanizeit.org/images/uploads/pdfs/ComparisonofContinuousSheetHot-DipGalv.pdf
If you are describing exposed structural framing on the exterior wall (such as roof screen support framing or mechanical supports) you would be better suited with hot dip galvanizing after fabrication to ASTM A123 Standard Specification for Zinc (Hot-Dip Galvanized) Coatings on Iron and Steel Products. This specification covers the requirements for zinc coating (galvanizing) by the hot-dip process on iron and steel products made from rolled pressed and forged shapes, castings, plates, bars, and strips.
The coating thickness is also measured in terms of mass or oz/ft2, but is not a total of both sides. For example, a piece of steel with a section thickness of ¼” or greater will yield a MINIMUM coating thickness of 2.3 oz/ft2 or 3.9 mils. The coating thickness on any surface will be at least 5 times the thickness and service life of a G-90 coating. Once again, the service life will be a function of the environment that the coating is exposed to. The link below is a service life chart showing the coating life versus coating thickness for a hot dip galvanized ASTM A123 coating.
http://www.galvanizeit.org/images/uploads/publicationPDFs/TFM_web.pdf
or visit the zinc coating life predictor at; http://www.galvinfo.com:8080/zclp/
A 3.5 to 3.9 mil ASTM A123 galvanized coating, in an industrial environment can be expected to provide between 60 and 70 years of maintenance free service life.
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
December 2010
Q: "Is it possible to have too much galvanized coating on a steel part?"
Questions like this really get the Professor's gears turning. He always has the answer or knows where to find it. The answer to this question comes from his pal Dr. Galv at the American Galvanizers Association (AGA).
Answer:
"The specifications for hot-dip galvanizing do not set a maximum coating thickness, so there is no limit on the thickness of the hot-dip galvanized coating. However, there is a requirement the coating meet the “intended use of the product,” so if there are drips and runs in an area that will cause a fit problem in the part’s final assembly, then the drips and runs must be ground smooth for the part to be acceptable.
Although the specification does not limit the coating thickness, there are some practical limits on how thick the galvanized coating should be. When the coating exceeds 10 mils or 250 microns, the coating experiences significant stress during the cool down from galvanizing temperatures. The thermal contraction of steel progresses at a different rate than the thermal contraction of the intermetallic layers of the galvanized coating. This difference in contraction rates induces a stress at the interface between layers of the coating. The stress level induced depends on the thickness of the intermetallic layers. At normal coating thickness levels, the stress is minimal and there is no issue with the coating adherence. However, if the coating is thick then the stress can be significant, and this makes the coating susceptible to flaking."
Download the complete answer, with photographic illustration, from the AGA now at www.galvanizeit.org
What's Your Question?
Submit your technical galvanizing question now! We'll get back to you with the Professor's answer ASAP. It could be featured on this page or in our next e-newsletter. Think of the honor!
|
|
||||
|
||||
|
|